Polar or Nonpolar? A+ Cation Polarity Control in A2Ti(IO3)6(A = Li, Na, K, Rb, Cs, Tl) | Journal of the American Chemical Society

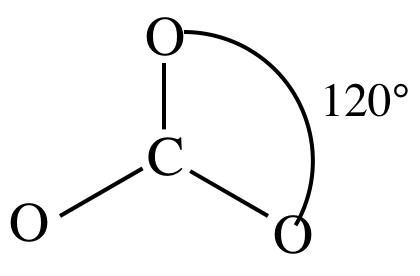
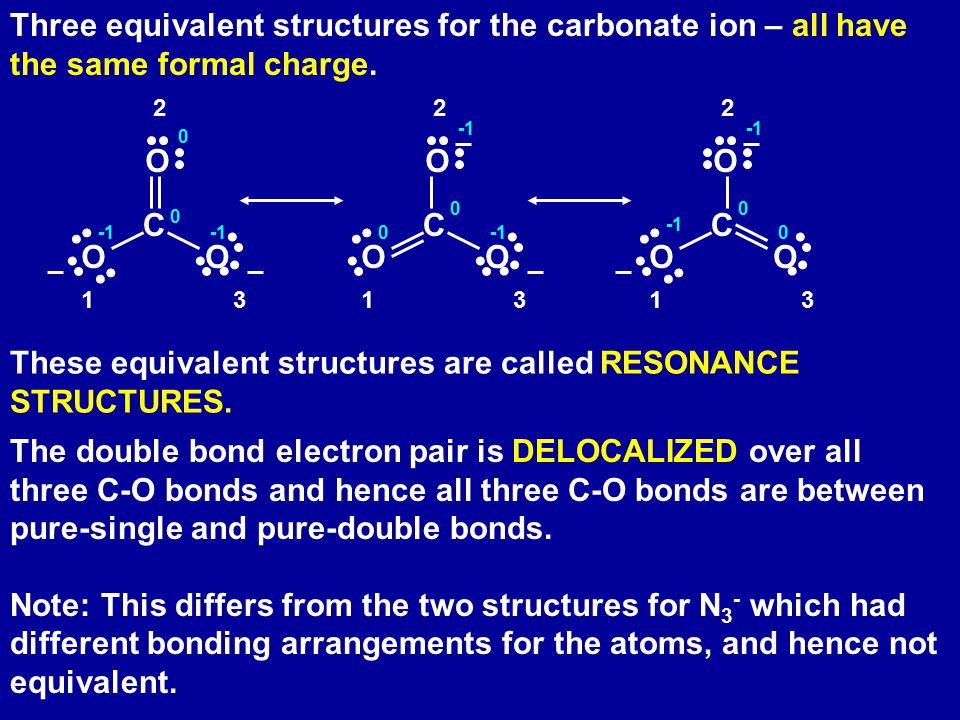
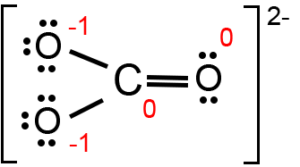
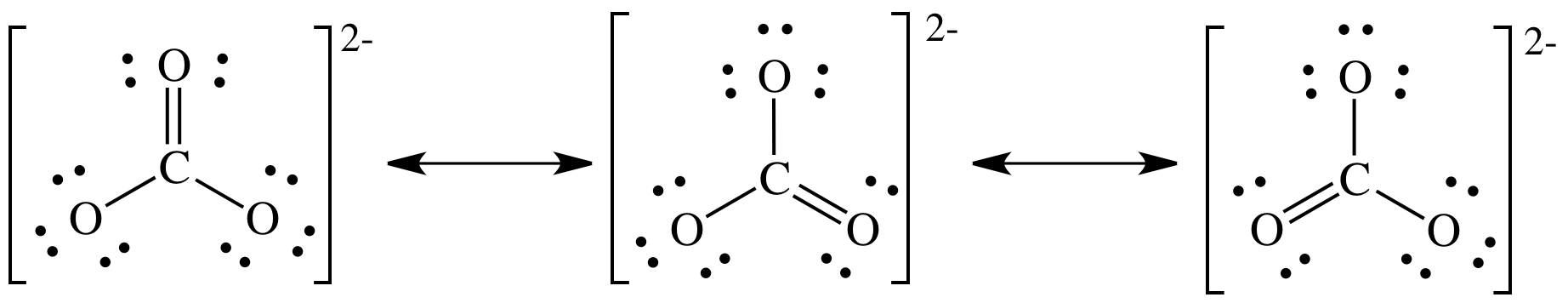
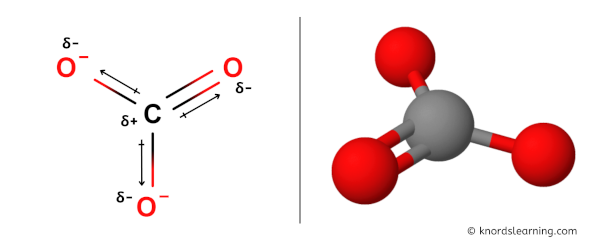
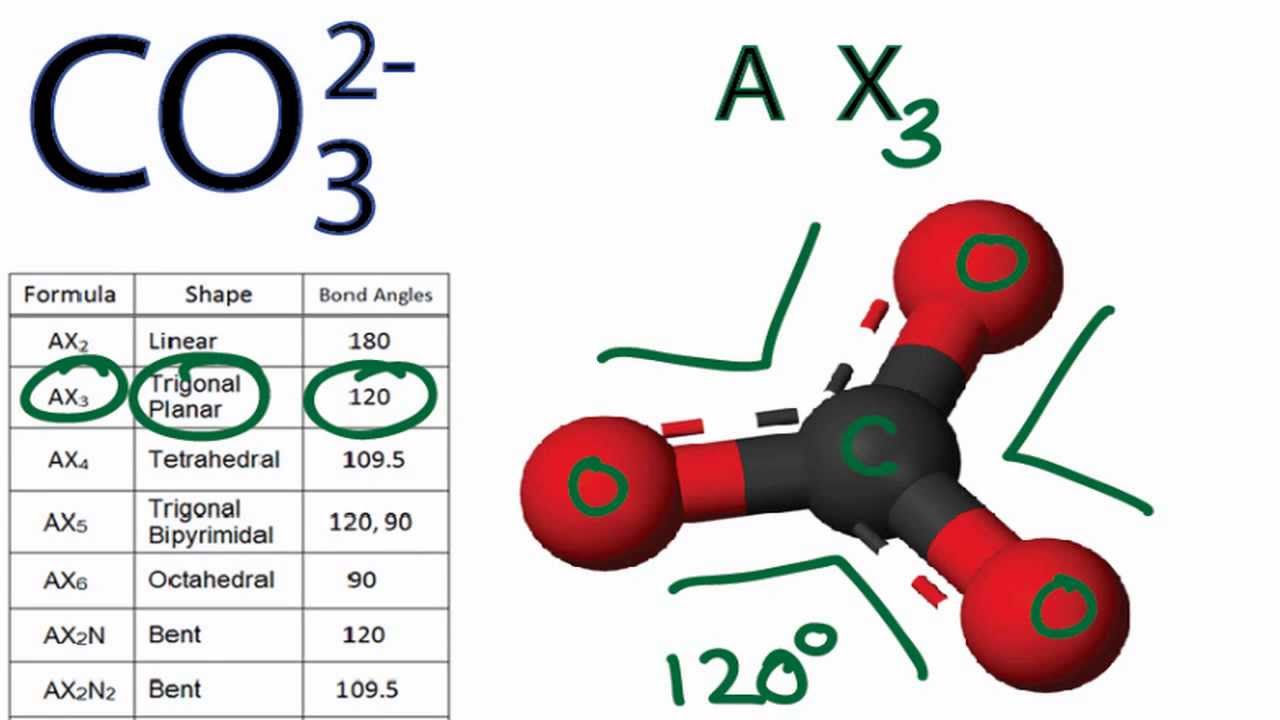
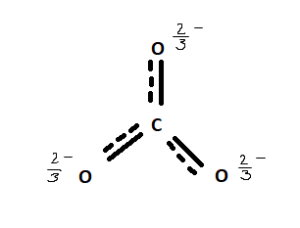
Regarding the carbonate ion, CO32-, which of the following statements is false? a. it has resonance b. it has formal charge only on its O atoms c. it has 24 valence electrons
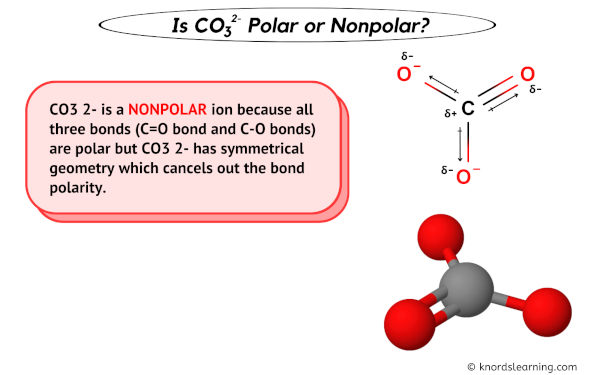
Can a molecule (like CO3 2-) be nonpolar even if it has two single bonds and one double bonds? - Quora