
The fourth line of the balmer series corresponds to electron transition between which energy levels? - Quora

Serie espectral de hidrógeno serie Balmer serie Lyman átomo de hidrógeno, energía, ángulo, texto png | PNGEgg

The fourth line of the balmer series corresponds to electron transition between which energy levels? - Quora

The fourth line of the Balmer series corresponds to the electronic transition between two orbits the H atom, Identify the orbits.

The fourth line of the balmer series corresponds to electron transition between which energy levels? - Quora

Serie espectral de hidrógeno serie Balmer serie Lyman átomo de hidrógeno, energía, ángulo, texto, electrón png | PNGWing

The fourth line of the balmer series corresponds to electron transition between which energy levels? - Quora

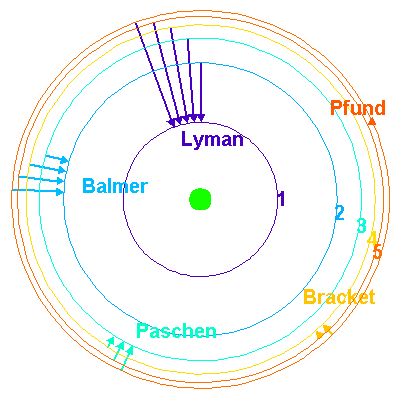






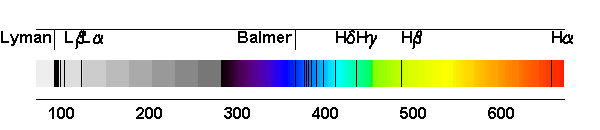









![What is the wave number of 4^th line in Balmer series of hydrogen spectrum? [R = 1,09,677cm^-1] What is the wave number of 4^th line in Balmer series of hydrogen spectrum? [R = 1,09,677cm^-1]](https://i.ytimg.com/vi/J2bcy4E6c6c/maxresdefault.jpg)